 
When the electrons eventually strike the atoms in the glass at the right end of the spectrometer, they excite electrons intrinsic to the atoms to a higher energy state. The electrons are attracted towards the anode where they are sharpened to a beam called a cathode ray and continue their way to the electric field plates and electromagnet where they are deflected. These electrons collide with other gas molecules and knock more electrons off them, creating a cascade of ions and electrons called a Townsend discharge. Instead of producing electrons from the cathode via a heated filament, the ioniser works on the principle of a ‘cold cathode’ where a high potential difference maintained between the cathode and anode accelerates the free electrons present in the trace amount of air. Consequently, free electrons are present amongst the trace amount of air molecules in the ioniser. The ioniser on the left of the spectrometer is not a complete vacuum but filled with a trace amount of air molecules, which are always in equilibrium between their neutral and ionic forms due to natural occurring processes like photoionisation. Using the spectrometer, he determined the charge-to-mass ratio of one of these electrons.  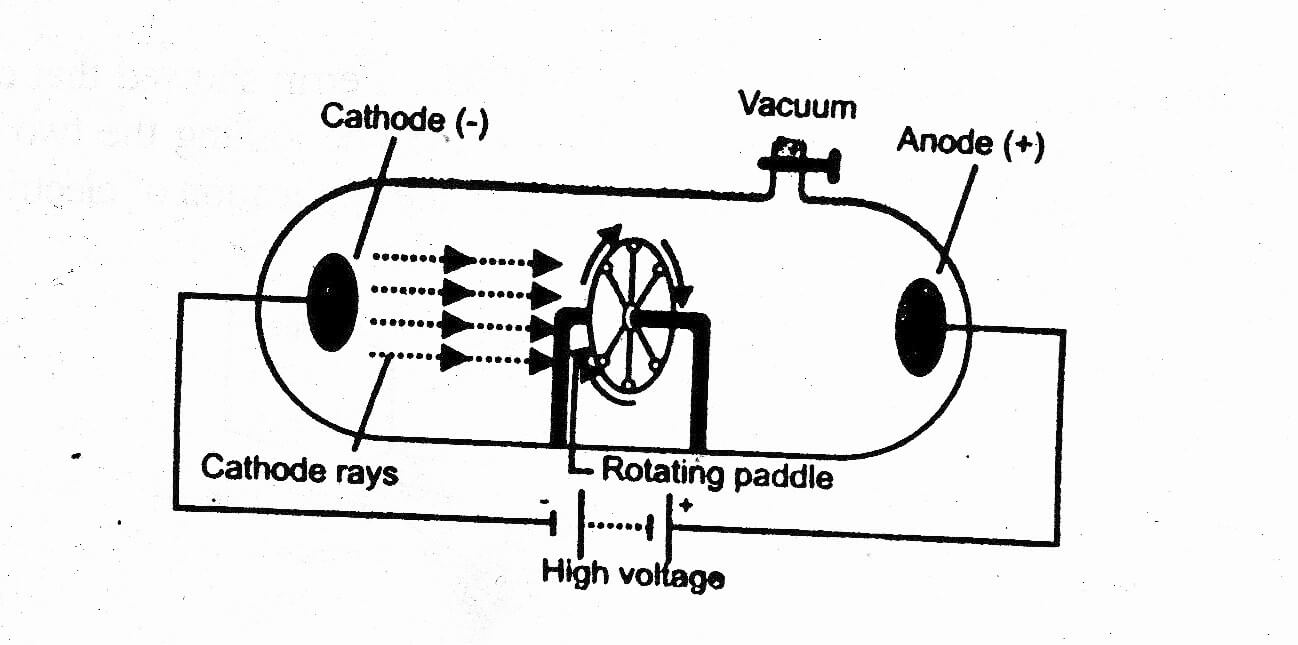
Thomson, an English physicist, constructed one of the earliest mass spectrometers (see below diagram) and demonstrated in an experiment in 1897 that atoms are made of subatomic particles called electrons. Mass spectrometry is an analytical technique that makes use of an instrument called a mass spectrometer to identify and quantify ions based on their mass-to-charge ratios. J. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
